Photo: The clipper ship Taifun slowly rusting in St George's Harbour. Credit: Montoya Lawrence, via Photos of Bermuda group.
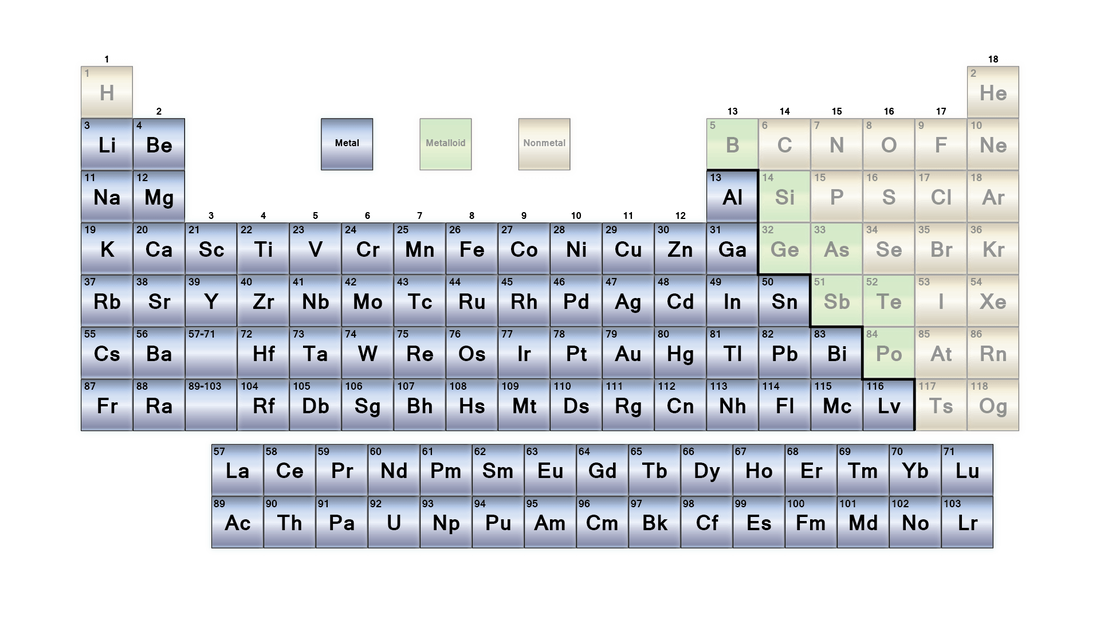
This unit looks at a major aspect of chemistry, the world of salts and carbonates. This unit deviates from the text book slightly as we are emphasizing the pure chemistry and not looking too much at the science of materials and rocks. Metal elements make up the majority of the periodic table and most will react to form substances such as salts and carbonates.
This unit looks at a major aspect of chemistry, the world of salts and carbonates. This unit deviates from the text book slightly as we are emphasizing the pure chemistry and not looking too much at the science of materials and rocks. Metal elements make up the majority of the periodic table and most will react to form substances such as salts and carbonates.
|
Cover photo: The rusting wreck of the barque Taifun in Convict Bay, St Georges. The iron of the ship is slowly reacting with the oxygen in the air to form the compound iron oxide - rust. She arrived after a storm in 1921 with a cargo of clay. When she sank the clay solidified and preserved much of the wreck. The upper decks have deteriorated a lot in the last 20 years or so. For many years she acted as a wharf for the boatyard, before becoming too unstable and dangerous. By the look of the only surviving photo of her under sail, she had long suffered from the all too common problem of a lack of preventative maintenance to isolate the iron from the oxygen, i.e. painting).
|
A - Metals
Objectives:
- Describe how properties of materials dictate their uses
- Describe the properties of metal elements
- Explain how decisions can be made about which metals to use in a given situation.
What makes a metal a metal? We all know whether something is a metal or not, but how exactly?
a) metals are shiny - well.... not all of them as some will very quickly tarnish
b) metals are all silvery - well.... not copper or gold
c) metals all conduct electricity - true, but then so does carbon, which isn't a metal
d) metals are strong - well.... sodium and potassium are only as hard as cheese
e) metals are all solid - well.... mercury isn't and gallium will melt in your hand
Hum...
a) metals are shiny - well.... not all of them as some will very quickly tarnish
b) metals are all silvery - well.... not copper or gold
c) metals all conduct electricity - true, but then so does carbon, which isn't a metal
d) metals are strong - well.... sodium and potassium are only as hard as cheese
e) metals are all solid - well.... mercury isn't and gallium will melt in your hand
Hum...
B- Metal Compounds
Objectives:
- Understand the neutralization reaction
- Identify changes that show a chemical reaction has taken place
- Know the difference between salts, carbonates and oxides.
Salts are a compound of a metal and a non-metal. The most common is 'table salt' which is sodium chloride \(\text{NaCl}\). Other salts form families such as chlorides, sulphides, iodides, sulphates, carbonates, nitrates etc. They can be made by reacting a metal oxide with an acid.
\[copper \, oxide + sulphuric \, acid \rightarrow copper\, sulphate + water\]
\[\text{CuO}+\text{H}_{2}\text{SO}_{4}\rightarrow \text{CuSO}_{4}+\text{H}_{2}\text{O}\]
\[sodium \,hydroxide + hydrochloric\, acid \rightarrow sodium \,chloride + water\]
\[\text{NaOH}+\text{HCl}\rightarrow \text{NaCl}+\text{H}_{2}\text{O}\]
or by displacement reactions with a more reactive metal. An example:
\[lead \,nitrate + potassium \,iodide \rightarrow potassium \,nitrate + lead\, iodide\] (bright yellow insoluble salt)
Or even by a direct reaction (scary!). See the youtube clip below. Commentary gets a bit technical, but the reaction is fun. Dangerous as chlorine gas is very toxic.
\[copper \, oxide + sulphuric \, acid \rightarrow copper\, sulphate + water\]
\[\text{CuO}+\text{H}_{2}\text{SO}_{4}\rightarrow \text{CuSO}_{4}+\text{H}_{2}\text{O}\]
\[sodium \,hydroxide + hydrochloric\, acid \rightarrow sodium \,chloride + water\]
\[\text{NaOH}+\text{HCl}\rightarrow \text{NaCl}+\text{H}_{2}\text{O}\]
or by displacement reactions with a more reactive metal. An example:
\[lead \,nitrate + potassium \,iodide \rightarrow potassium \,nitrate + lead\, iodide\] (bright yellow insoluble salt)
Or even by a direct reaction (scary!). See the youtube clip below. Commentary gets a bit technical, but the reaction is fun. Dangerous as chlorine gas is very toxic.
|
\[sodium + chlorine \rightarrow sodium \,chloride\]
\[2\text{Na}+\text{Cl}_{2}\rightarrow 2\text{NaCl}\] |
|
Lots of interesting questions about salts:
- is there a pattern to their colours?
- is there a pattern to their solubility?
- where did the salt in the ocean come from?
- how much salt is in the ocean?
- is the salinity of oceans constant?
- how does the amount of salt that can be dissolved vary with temperature?
- how does salt affect the freezing point of water?
- what are various salts used for?
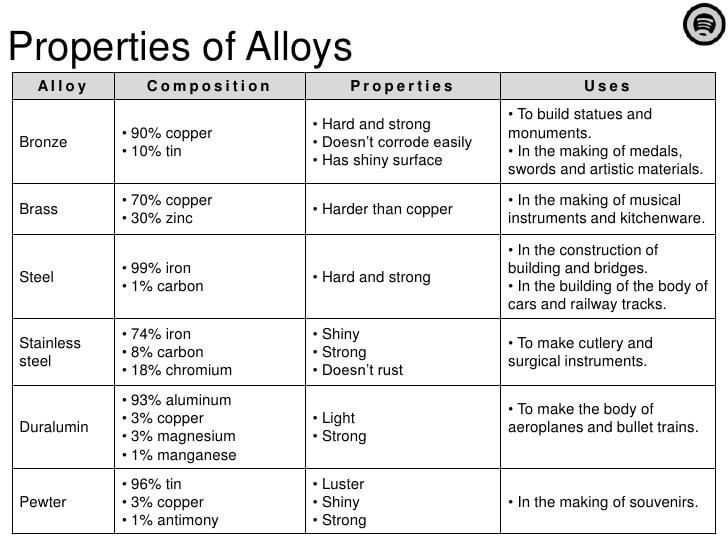
C - Metal Alloys
Objectives:
- Describe how alloys make metals more useful
- Use the particle model to explain why an alloy can be stronger than a pure metal
- Recall the ingredients of some common alloys
Common alloys:
- BRASS - copper and zinc
- BRONZE - copper and tin
- AMALGAM - mercury and either silver or gold
- PEWTER - tin and copper
- STERLING SILVER - silver and copper
- STEEL - iron and carbon
- WHITE GOLD - gold and silver
- STAINLESS STEEL - varies!
LAB WORK - Making solder from melting a mixture of lead and tin pieces.