|
Oceanography >> Seawater >> Measuring Salinity
|
2.2 - Measuring Salinity
- To be able to measure the salinity of seawater using changes of density or conductivity
- To be able to find salinity charts that have been measured from space
Salinity is one of the two key variables of the water that makes up the ocean, the other being temperature. Therefore it is essential to be able to measure it accurately.
Definition: salinity is the number of grams of dissolved solids in a kilogram of seawater. As the amount of salt dissolved is relatively small, around \(35\,\text{g}\), it is customary to measure in parts per thousand, which is written in a similar manner to the percentage symbol - \( 35\,\unicode{x2030}\). The other more precise unit is the Practical Salinity Unit \( \text{(PSU)}\), which to all intents and purposes is the same thing.
There are three methods that we can use in the lab to measure salinity. We are going to use our salt water samples that we made last before to test and calibrate our techniques before using it on some actual seawater.
Definition: salinity is the number of grams of dissolved solids in a kilogram of seawater. As the amount of salt dissolved is relatively small, around \(35\,\text{g}\), it is customary to measure in parts per thousand, which is written in a similar manner to the percentage symbol - \( 35\,\unicode{x2030}\). The other more precise unit is the Practical Salinity Unit \( \text{(PSU)}\), which to all intents and purposes is the same thing.
There are three methods that we can use in the lab to measure salinity. We are going to use our salt water samples that we made last before to test and calibrate our techniques before using it on some actual seawater.
LAB 2.2 - Evaporation
As the salinity is due to dissolved solids, mainly salt, in the water, removing the water will leave behind the solids. As boiling away \(1.0\,\text{kg}\) of water is rather tedious, we can use a small amount, say \(50\, \text{g}\) instead. You will need to carefully weigh the evaporating basin before and after. Each group can do a different sample of water and we can discuss the results as a class.
As the salinity is due to dissolved solids, mainly salt, in the water, removing the water will leave behind the solids. As boiling away \(1.0\,\text{kg}\) of water is rather tedious, we can use a small amount, say \(50\, \text{g}\) instead. You will need to carefully weigh the evaporating basin before and after. Each group can do a different sample of water and we can discuss the results as a class.
LAB 2.3 - Measuring the Density
Fundamentally, seawater is denser than freshwater as the sodium and chloride ions get between the spaces between the water molecules. Upshot is that the volume does not change by an appreciable amount when salt is dissolved into the water. The more salt there is dissolved in the water the denser it becomes. So, \(1.0\,\text{l}\) of seawater will weigh slightly more than \(1.0\,\text{l}\) of freshwater. However, it is NOT quite this straightforward! As the temperature of water also affects its density slightly we need to ensure that the temperature remains constant. As the lab is about \(20^{\circ}\text{C}\), we will use that as our control temperature.
Method A - Measure out exactly \(100.0\,\text{ml}\) of each sample of water and weigh it. Do not forget to account for the mass of the measuring cylinder! Use the equation below to calculate the density: \[\rho=\frac{m}{V}\]
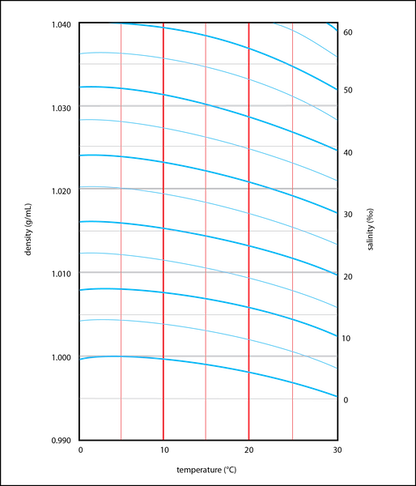
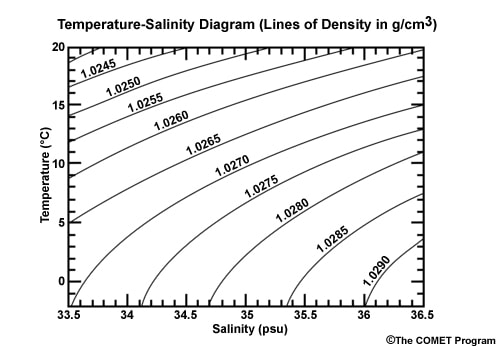
The salinity can then be determined from the graphs below. The one on the right is more precise, but has a smaller range.
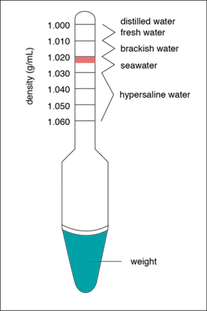
Method B - Using a HYDROMETER we can determine both the density and salinity from the buoyancy of the hydrometer.
Fundamentally, seawater is denser than freshwater as the sodium and chloride ions get between the spaces between the water molecules. Upshot is that the volume does not change by an appreciable amount when salt is dissolved into the water. The more salt there is dissolved in the water the denser it becomes. So, \(1.0\,\text{l}\) of seawater will weigh slightly more than \(1.0\,\text{l}\) of freshwater. However, it is NOT quite this straightforward! As the temperature of water also affects its density slightly we need to ensure that the temperature remains constant. As the lab is about \(20^{\circ}\text{C}\), we will use that as our control temperature.
Method A - Measure out exactly \(100.0\,\text{ml}\) of each sample of water and weigh it. Do not forget to account for the mass of the measuring cylinder! Use the equation below to calculate the density: \[\rho=\frac{m}{V}\]
The salinity can then be determined from the graphs below. The one on the right is more precise, but has a smaller range.
Method B - Using a HYDROMETER we can determine both the density and salinity from the buoyancy of the hydrometer.
LAB 2.4 - Conductivity of Water
Measuring the salinity of water either by evaporation or density can only really be successfully performed in the lab. This is not helpful for automated instruments on robots, CTDs or moorings. We need something that can be converted into an electrical signal somehow. It was known from chemistry that salt solutions conduct electricity. The greater the concentration of the solution the greater the ability of the solution to conduct. So, in theory, we can stick a couple of electrodes into the water and measure the current with an ammeter and bingo! But... with all things in science we need to be careful. The amount of current conducted through the water depends on many variables as well as just the salinity:
Measuring the salinity of water either by evaporation or density can only really be successfully performed in the lab. This is not helpful for automated instruments on robots, CTDs or moorings. We need something that can be converted into an electrical signal somehow. It was known from chemistry that salt solutions conduct electricity. The greater the concentration of the solution the greater the ability of the solution to conduct. So, in theory, we can stick a couple of electrodes into the water and measure the current with an ammeter and bingo! But... with all things in science we need to be careful. The amount of current conducted through the water depends on many variables as well as just the salinity:
- voltage of battery
- size of electrodes
- separation of electrodes
- temperature of the solution
- type of electrodes
- type of solution (although we will only consider sodium chloride)
Other Resources
Surprisingly it is possible to measure the surface salinity from space. The NASA satellite Aquarius did this a few years ago. I am not sure how it was done though!
https://www.nasa.gov/mission_pages/aquarius/overview/index.html
https://aquarius.oceansciences.org/cgi/science.cgi
https://www.nasa.gov/mission_pages/aquarius/overview/index.html
https://aquarius.oceansciences.org/cgi/science.cgi