This unit starts to introduce the quantitative aspect of chemistry, with the main objectives being to understand that concept of conservation of mass, balancing chemical equations (extension work) and an understanding of fuels and the combustion reaction.
A - Chemical Formulae
Objectives:
- To be able to list the elements that make up any given compound based on its chemical formula
- To be able to list the numbers of each type of atom in any given molecule based on its chemical formula
Chemical formulae may look odd, but they tell you the types and number of atoms that make up the molecule of an element or compound. Some also highlight the structure of the molecule. For example that the oxygen atom in ethanol is located between the carbon and a single hydrogen atom, or that there are two carbonate clusters that are bonded to a metal.
|
methane - \(\text{CH}_4\) - this has 1 x carbon atom bonded to 4 x hydrogen atoms.
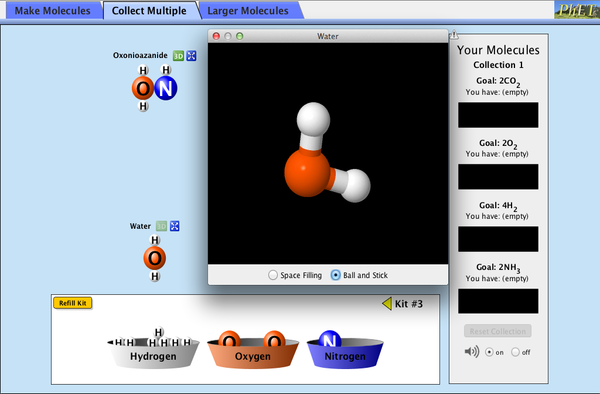
sodium nitrate - \(\text{NaNO}_3\) - has 1 x sodium atom, 1 x nitrogen atom and 3 x oxygen atoms. (Note - the ...ate ending on a compound generally means 3 or 4 oxygen atoms. Activity: run the simulation opposite and build a variety of molecules. |
B - Rates of Reaction
Objectives:
- Explain some of the variables that can increase the speed of a chemical reaction
Chemical reactions occur when the molecules collide and interact. Chemical reactions can go slowly or very fast. There are a number of factors that can increase the speed of a chemical reaction:
- temperature - increasing the temperature has a very pronounced effect on its speed. Raising the temperature increases the speed of the particles, so that they collide more often and more vigorously.
- concentration - increasing the concentration of a reactant (e.g. acid) rapidly increases its speed. This is because there are less inert particles getting in the way of reactant collisions.
- Surface area - increasing the surface area to volume ratio by (say) crushing it into a powder or melting it, exposes more of the reactants to contact with the other reactant, thereby speeding up the reaction.
- Stirring it - stirring the mixture again increases the chance and rate of collisions. Any reacted chemicals that have produced new molcules are cleared away, therefore increasing the rate of new reactants to collide.
|
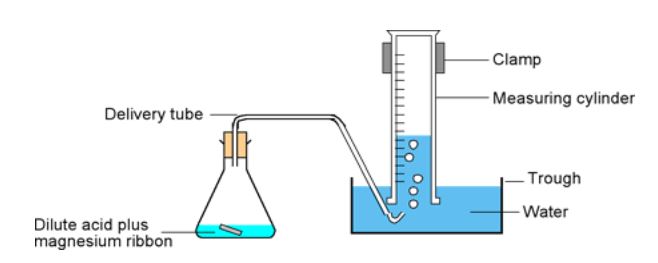
LAB WORK: we will investigate some of the factors that increase the rate of a chemical reaction. We use the set up shown and react magnesium and hydrochloric acid to produce hydrogen gas. The students time how long it takes to collect \(10 \,\text{ml}\) of hydrogen. Variables include: the concentration and temperature of the acid. Other factors that we do not investigate are: surface area of the reactant and stirring, so these are controlled.
|
C - Mass in Reactions
Objectives:
- Explain why mass is conserved in a chemical reaction
- Explain why some reactions seem to gain/lose mass
- Extension: be able to balance simple chemical equations
This is fundamental - the mass of the products must equal the mass of the reactants. In chemistry, no atoms are created or destroyed. They are rearranged. This is similar to Lego - the total bricks before and after must be the same, none are magically created out of thin air or vanish.
However, it often appears during experiments that the mass before is NOT equal to the mass after. This is because we can't weigh a gas! If oxygen is pulled out of the air to bond with (say) magnesium to produce magnesium oxide, it appears to have gained mass, but that is because we haven't weighed the oxygen before the reaction. Similarly if a reaction produces a gas that is allowed to escape into the atmosphere, the reaction appears to have lost mass.
For the more advanced students: this is shown in chemical equations by 'balancing' them. The coefficient is changed to ensure that the number of atoms going in must equal the number coming out of the reaction. This is a key part of chemistry at IGCSE or beyond.
eg - the burning of hydrogen to produce water:
Word equation is:
\[ hydrogen+oxygen \rightarrow water\]
The chemical equation (remembering that both hydrogen and oxygen gases are diatomic and exist as pairs) is:
\[\text{H}_2+\text{O}_2 \rightarrow\text{H}_{2}\text{O}\]
But this is incorrect as there are 2 x oxygens in the reactants and only 1 x oxygen in the product. An oxygen atom cannot just vanish. We can't just change the compound to read \(\text{H}_2\text{O}_2\) as that is hydrogen peroxide, which is most definitely NOT water! So we balance by changing the numbers of the molecules.
\[2\text{H}_2+\text{O}_2\rightarrow 2\text{H}_2\text{O}\]
On a practical, this means that we need twice the amount of hydrogen than we need oxygen to make water. This is how they figured all the formulae out all those years ago!
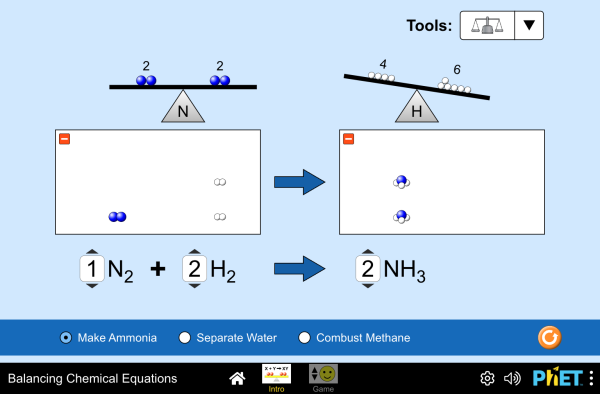
Activity - Use the PhET simulation below to balance equations.
However, it often appears during experiments that the mass before is NOT equal to the mass after. This is because we can't weigh a gas! If oxygen is pulled out of the air to bond with (say) magnesium to produce magnesium oxide, it appears to have gained mass, but that is because we haven't weighed the oxygen before the reaction. Similarly if a reaction produces a gas that is allowed to escape into the atmosphere, the reaction appears to have lost mass.
For the more advanced students: this is shown in chemical equations by 'balancing' them. The coefficient is changed to ensure that the number of atoms going in must equal the number coming out of the reaction. This is a key part of chemistry at IGCSE or beyond.
eg - the burning of hydrogen to produce water:
Word equation is:
\[ hydrogen+oxygen \rightarrow water\]
The chemical equation (remembering that both hydrogen and oxygen gases are diatomic and exist as pairs) is:
\[\text{H}_2+\text{O}_2 \rightarrow\text{H}_{2}\text{O}\]
But this is incorrect as there are 2 x oxygens in the reactants and only 1 x oxygen in the product. An oxygen atom cannot just vanish. We can't just change the compound to read \(\text{H}_2\text{O}_2\) as that is hydrogen peroxide, which is most definitely NOT water! So we balance by changing the numbers of the molecules.
\[2\text{H}_2+\text{O}_2\rightarrow 2\text{H}_2\text{O}\]
On a practical, this means that we need twice the amount of hydrogen than we need oxygen to make water. This is how they figured all the formulae out all those years ago!
Activity - Use the PhET simulation below to balance equations.
D - Energetic Reactions
- Understand the chemistry of combustion
- Recall the compounds made when fuels burn
- Describe the advantages and disadvantages of different fuels
Many chemical reactions are EXOTHERMIC, which means that they release heat energy. The most familiar is the combustion (burning) reaction that is used almost every day!
The general reaction is:
\[fuel+oxygen \rightarrow oxides\]
Most fuels contain the elements hydrogen and carbon (hydrocarbons and/or carbohydrates).
\[hydrogen+oxygen \rightarrow water\]
\[carbon+oxygen \rightarrow carbon\,dioxide\]
So the general equation becomes:
\[fuel+oxygen \rightarrow water+carbon\,dioxide\]
For example, the burning of the simplest hydrocarbon (methane) is:
\[\text{CH}_4+2\text{O}_2 \rightarrow \text{CO}_2+2\text{H}_2\text{O}\]
Note - if there is NOT ENOUGH oxygen for the reaction to be completed, we get incomplete combustion. The carbon does not bond with the right amount of oxygen and some pure carbon is formed (soot and smoke) and carbon monoxide \(\text{CO}\), which is a toxic gas. A tell tale sign of incomplete combustion is a yellow flame. This is why we need adequate ventilation when we burn candles and wood.
The general reaction is:
\[fuel+oxygen \rightarrow oxides\]
Most fuels contain the elements hydrogen and carbon (hydrocarbons and/or carbohydrates).
\[hydrogen+oxygen \rightarrow water\]
\[carbon+oxygen \rightarrow carbon\,dioxide\]
So the general equation becomes:
\[fuel+oxygen \rightarrow water+carbon\,dioxide\]
For example, the burning of the simplest hydrocarbon (methane) is:
\[\text{CH}_4+2\text{O}_2 \rightarrow \text{CO}_2+2\text{H}_2\text{O}\]
Note - if there is NOT ENOUGH oxygen for the reaction to be completed, we get incomplete combustion. The carbon does not bond with the right amount of oxygen and some pure carbon is formed (soot and smoke) and carbon monoxide \(\text{CO}\), which is a toxic gas. A tell tale sign of incomplete combustion is a yellow flame. This is why we need adequate ventilation when we burn candles and wood.
LAB WORK - you will be designing and carrying out an experiment into the amount of energy released by burning \(1.0\,\text{g}\) of a variety of fuels. Fuels with be various alcohols.
|
|
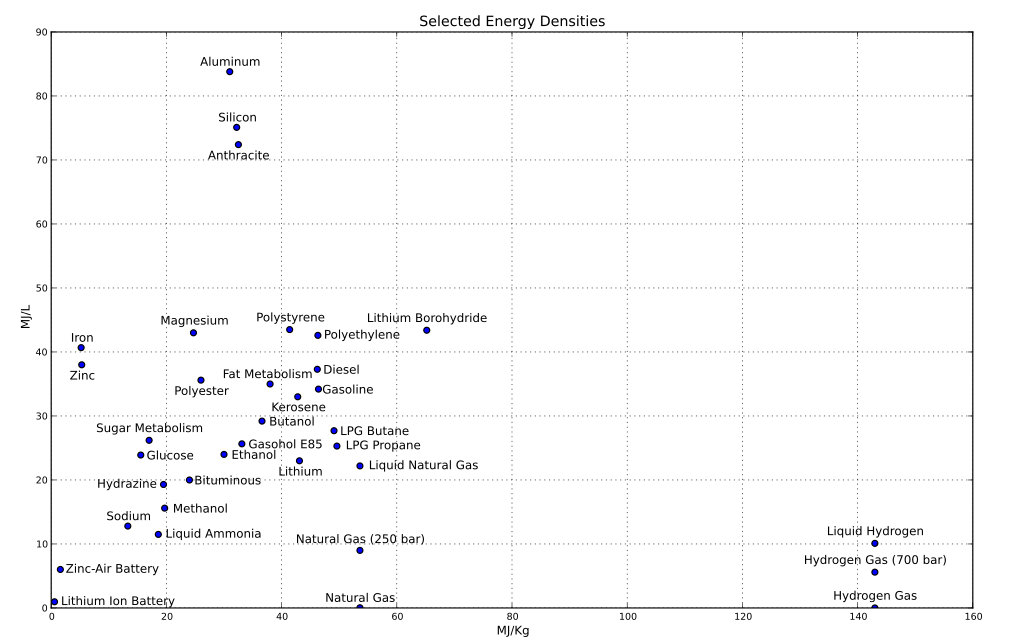
Scatter plot of the energy densities of various fuels. In terms for bang for mass, hydrogen is by far the best. However, in terms of bang for volume, you need aluminium. Which is quite hard to burn in air, but in a high pressure oxygen environment and in powered form it goes bang pretty easily. I can't remember where I got this image from.
|